Define Units
A unit is a standard measure of a physical quantity. It tells us in what terms the measurement is expressed.
Unit Examples
The unit of length is the meter, which has the abbreviation (m). The unit of mass is the kilogram, which has the abbreviation (kg). The unit of time is the second, which has the abbreviation (s).
Define Dimensions
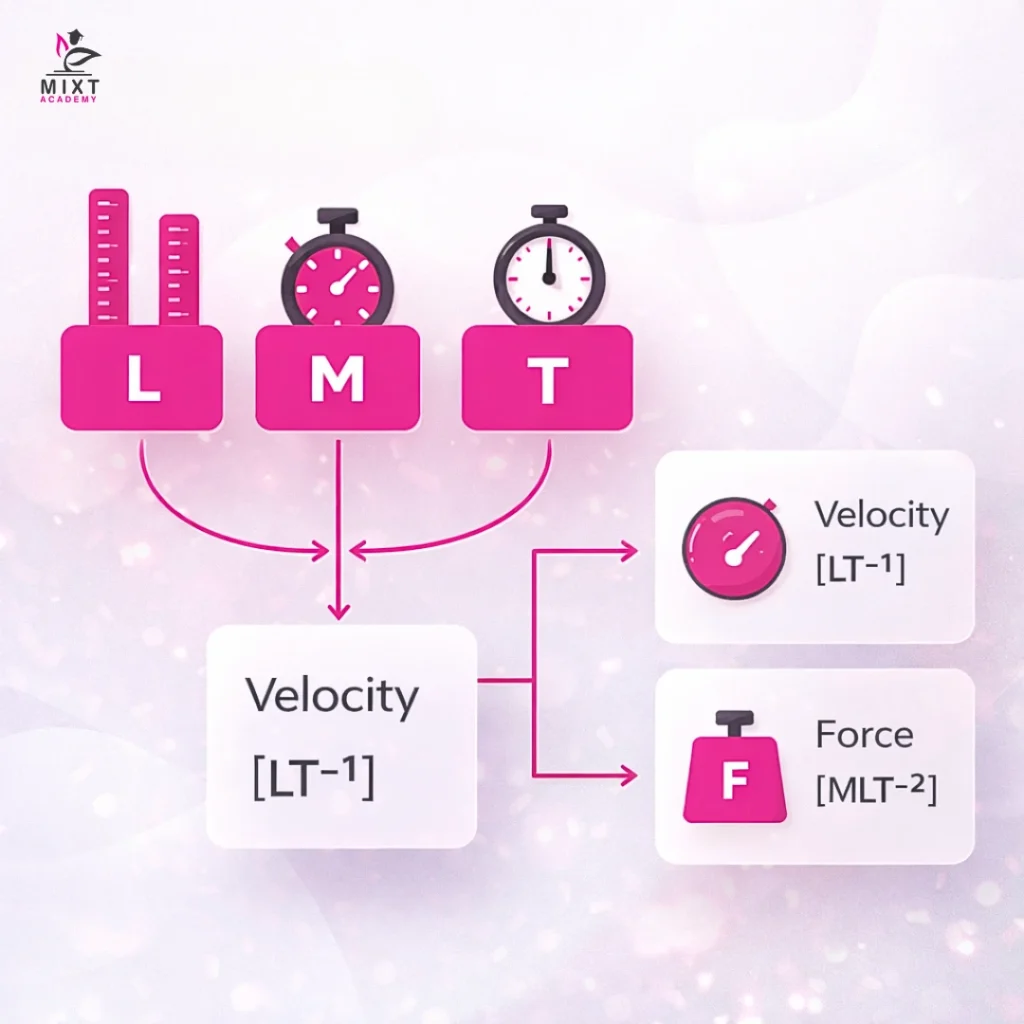
Dimensions define the characteristics of a physical quantity using basic units, such as length (L), mass (M), and time (T). The dimension of velocity shows its representation through [LT^(-1)] while the dimension of force shows its representation through [MLT^(-2) ].
Need Help Understanding Physical Quantities, Their Units, and Dimensions?
Mixt Academy is here to help you achieve your academic goals.
Dimensional Analysis and Related Concepts
Dimensional Formula
The dimensional formula shows how a derived quantity relates to fundamental units through the specific powers of those units. The dimensional formula for the derived quantity Q is Q = M^a L^b T^c; the term M^a L^b T^c is its dimensional formula, where the exponents a, b, and c are called the dimensions of the quantity.
Dimensional Constants
Physical quantities that have fixed values and possess dimensions are called dimensional constants. Examples include the gravitational constant G, Planck’s constant h, the universal gas constant R, and the speed of light in a vacuum c.
Dimensionless Quantities

Dimensionless quantities refer to physical, chemical, or mathematical values that lack measurement units and possess a net dimension of one.
- Without Units: These quantities have no dimensions and no units; they are pure numbers, such as π, e, and trigonometric functions like sinθ, cosθ, and tanθ.
- With Units: Some dimensionless quantities are expressed with special units, for example, angular displacement measured in radians or Joule’s constant expressed as joules per calorie.
Dimensional Variables
Quantities with dimensions whose values can vary are called dimensional variables. Common examples include velocity, acceleration, force, work, and power.
Dimensionless Variables
Dimensionless variables are physical quantities that have no dimensions and whose values can vary. Examples include specific gravity, refractive index, coefficient of friction, and Poisson’s ratio.
Looking for Trusted Online Revision Material on Dimensional Analysis?
Stop memorising, start understanding and get quality revision material.
Law of Homogeneity of Dimensions

For any physically meaningful equation relating physical quantities, the dimensions of every term on both sides must be identical.
Conversion of Numerical Values Between Unit Systems
Consider a physical quantity with dimensions a, b, and c corresponding to length (L), mass (M), and time (T), respectively. If n_1is the numerical value of Qin a system with fundamental units L_1, M_1, and T_1, and n_2 is its numerical value in another system with fundamental units L_2, M_2, and T_2, then the values are related by:
n2 = n1 (L1⁄L2)a (M1⁄M2)b (T1⁄T2)c
Useful Unit Conversions in Physics
Pressure Conversions:
- 1 bar = 10⁶ dyne/cm² = 10⁵ N/m² = 10⁵ Pa
- 76 cm of Hg ≈ 1.013 × 10⁶ dyne/cm² = 1.013 × 10⁵ Pa ≈ 1.013 bar
- 1 torr (1 mm Hg) = 1.333 × 10³ dyne/cm² ≈ 1.333 mbar
Force and Mass:
- 1 dyne = 10⁻⁵ N
- 1 kgf (kilogram-force) = 9.81 N
Energy and Work:
- 1 calorie (cal) ≈ 4.184 joule (J)
- 1 erg = 10⁻⁷ J
- 1 electron volt (eV) = 1.602 × 10⁻¹⁹ J
- 1 kilowatt-hour (kWh) = 3.6 × 10⁶ J
Power:
- 1 horsepower (H.P.) = 746 W
Speed:
- 1 km/h = 5/18 m/s ≈ 0.2778 m/s
Essential Physical Constants

Fundamental Speeds:
- Velocity of light in vacuum: c=3×10^8 m/s
- Velocity of sound in air at STP: v=331 m/s
Gravitational & Mechanical Constants:
- Acceleration due to gravity: g=9.81 m/s²
- Universal gravitational constant: G=6.67×10^−11 N·m²/kg²
- Mechanical equivalent of heat: J=4.186 J/cal
Atomic & Molecular Constants:
- Avogadro number: NA=6.022 × 10^23 mol⁻¹
- Atomic mass unit: 1 u=1.66 × 10^−27 kg
- Quantum of charge: e=1.602×10⁻¹⁹ C
Thermal & Radiation Constants:
- Boltzmann constant: k=1.381×10^−23 J/K
- Stefan-Boltzmann constant: σ=5.67×10−8 W/m²·K⁴
- Absolute zero: 0 K = −273.15 °C
Gas & Pressure Constants:
- Universal gas constant: R=8.314 J/mol·K
- One atmosphere: 1 atm = 76 cm Hg = 1.013 × 10⁵ Pa
Electromagnetic Constants:
- Permittivity of free space: ε_0=8.854×10^−12
- Permeability of free space: μ_0=4π×10−7 H/m
- Planck’s constant: h=6.626×10^−34 J·s
Density Constants:
- Density of water at 4 °C: ρ=1000 kg/m³ = 1 g/cm³
- Density of air at STP: ρ=1.293 kg/m³
Derived SI Units with Special Names
The following are commonly used derived SI units that have been assigned special names:
| Physical Quantity | SI Unit | Symbol |
|---|---|---|
| Frequency | hertz | Hz |
| Energy | joule | J |
| Force | newton | N |
| Power | watt | W |
| Pressure | pascal | Pa |
| Electric charge (quantity of electricity) | coulomb | C |
| Electric potential difference / EMF | volt | V |
| Electric resistance | ohm | Ω |
| Electric conductance | siemens | S |
| Electric capacitance | farad | F |
| Magnetic flux | weber | Wb |
| Inductance | henry | H |
| Magnetic flux density | tesla | T |
| Illumination | lux | lx |
| Luminous flux | lumen | lm |
Dimensional Formulas of Common Physical Quantities

The dimensional formula of a physical quantity expresses it in terms of the fundamental quantities: Mass (M), Length (L), Time (T), Electric current (I), Temperature (θ), Luminous intensity (J), and Amount of substance (N). Derived quantities are written as products of these fundamentals raised to specific powers.
| Physical Quantity | Unit | Dimensional Formula |
|---|---|---|
| Acceleration (or gravity) | m/s² | LT⁻² |
| Angle / Angular displacement | rad | MoLoTo |
| Angular frequency | rad/s | T⁻¹ |
| Angular impulse (torque × time) | N·m·s | ML²T⁻¹ |
| Angular momentum (Iω) | kg·m²/s | ML²T⁻¹ |
| Angular velocity | rad/s | T⁻¹ |
| Area | m² | L² |
| Boltzmann constant | J/K | ML²T⁻²θ⁻¹ |
| Bulk modulus | Pa | ML⁻¹T⁻² |
| Calorific value | J/kg | L²T⁻² |
| Coefficient of linear/areal/volume expansion | °C⁻¹ or K⁻¹ | θ⁻¹ |
| Surface tension (force/length) | N/m | MT⁻² |
| Thermal conductivity | W/m·K | MLT⁻³θ⁻¹ |
| Viscosity | poise | ML⁻¹T⁻¹ |
| Compressibility (1/bulk modulus) | Pa⁻¹ | M⁻¹LT² |
| Density | kg/m³ | ML⁻³ |
| Displacement / Wavelength / Focal length | m | L |
| Electric capacitance (charge/potential) | F | M⁻¹L⁻²T⁴I² |
| Electric conductance | S | M⁻¹L⁻²T³I² |
| Electric conductivity | S/m | M⁻¹L⁻³T³I² |
| Electric charge | C | IT |
| Electric current | A | I |
| Electric dipole moment | C·m | LTI |
| Electric field strength (force/charge) | N/C | MLT⁻³I⁻¹ |
| Electric resistance | Ω | ML²T⁻³I⁻² |
| EMF / Electric potential | V | ML²T⁻³I⁻¹ |
| Energy | J | ML²T⁻² |
| Energy density | J/m³ | ML⁻¹T⁻² |
| Entropy | J/θ | ML²T⁻²θ⁻¹ |
| Force | N | MLT⁻² |
| Force constant / Spring constant | N/m | MT⁻² |
| Frequency | Hz | T⁻¹ |
| Gravitational potential | J/kg | L²T⁻² |
| Heat / Energy | J | ML²T⁻² |
| Illumination / Illuminance | lux | MT⁻³ |
| Impulse | N·s | MLT⁻¹ |
| Inductance | H | ML²T⁻²I⁻² |
| Intensity of gravitational field | N/kg | LT⁻² |
| Intensity of magnetisation | A/m | L⁻¹I |
| Joule’s constant | J/cal | MoLoTo |
| Latent heat | J/kg | L²T⁻² |
| Linear density | kg/m | ML⁻¹ |
| Luminous flux | lumen | ML²T⁻³ |
| Magnetic dipole moment | A·m² | L²I |
| Magnetic flux | Wb | ML²T⁻²I⁻¹ |
| Magnetic induction | T | MT⁻²I⁻¹ |
| Magnetic pole strength | A·m | LI |
| Modulus of elasticity | Pa | ML⁻¹T⁻² |
| Moment of inertia | kg·m² | ML² |
| Momentum | kg·m/s | MLT⁻¹ |
| Permeability of free space | H/m | MLT⁻²I⁻² |
| Permittivity of free space | F/m | M⁻¹L⁻³T⁴I² |
| Planck’s constant | J·s | ML²T⁻¹ |
| Poisson’s ratio | – | MoLoTo |
| Power | W | ML²T⁻³ |
| Pressure | Pa | ML⁻¹T⁻² |
| Pressure coefficient / Volume coefficient | °C⁻¹ | θ⁻¹ |
| Pressure head | m | MoLTo |
| Radioactivity | disintegrations/s | MoLoT⁻¹ |
| Ratio of specific heats | – | MoLoTo |
| Refractive index | – | MoLoTo |
| Resistivity | Ω·m | ML³T⁻³I⁻² |
| Specific conductance | S/m | M⁻¹L⁻³T³I² |
| Specific entropy | 1/K | M⁻¹L⁻²T²θ |
| Specific gravity | – | MoLoTo |
| Specific heat | J/kg·θ | L²T⁻²θ⁻¹ |
| Specific volume | m³/kg | M⁻¹L³ |
| Speed | m/s | LT⁻¹ |
| Stefan’s constant | W/m²·θ⁴ | MLT⁻³θ⁻⁴ |
| Strain | – | MoLoTo |
| Stress | Pa | ML⁻¹T⁻² |
| Surface energy density | J/m² | MT⁻² |
| Temperature | °C or θ | MoLoToθ |
| Temperature gradient | °C/m | MoL⁻¹Toθ |
| Thermal capacity | J/θ | ML²T⁻²θ⁻¹ |
| Time period | s | T |
| Torque / Moment of force | N·m | ML²T⁻² |
| Universal gas constant | J/mol·θ | ML²T⁻²θ⁻¹ |
| Universal gravitational constant | N·m²/kg² | M⁻¹L³T⁻² |
| Velocity | m/s | LT⁻¹ |
| Velocity gradient | s⁻¹ | T⁻¹ |
| Volume | m³ | L³ |
| Water equivalent | kg | MoLoTo |
| Work | J | ML²T⁻² |
Physical Quantities Sharing the Same Dimensional Formula
- Impulse and momentum
- Force and thrust
- Work, energy, torque, and moment of force
- Angular momentum, Planck’s constant, and rotational impulse
- Stress, pressure, modulus of elasticity, and energy density
- Force constant, surface tension, and surface energy
- Angular velocity, frequency, and velocity gradient
- Gravitational potential and latent heat
- Thermal capacity, entropy, universal gas constant, and Boltzmann constant
- Power and luminous flux
Too Many Constants to Remember?
We teach smart shortcuts and patterns so you don’t have to memorise everything blindly.
Applications of Dimensional Analysis
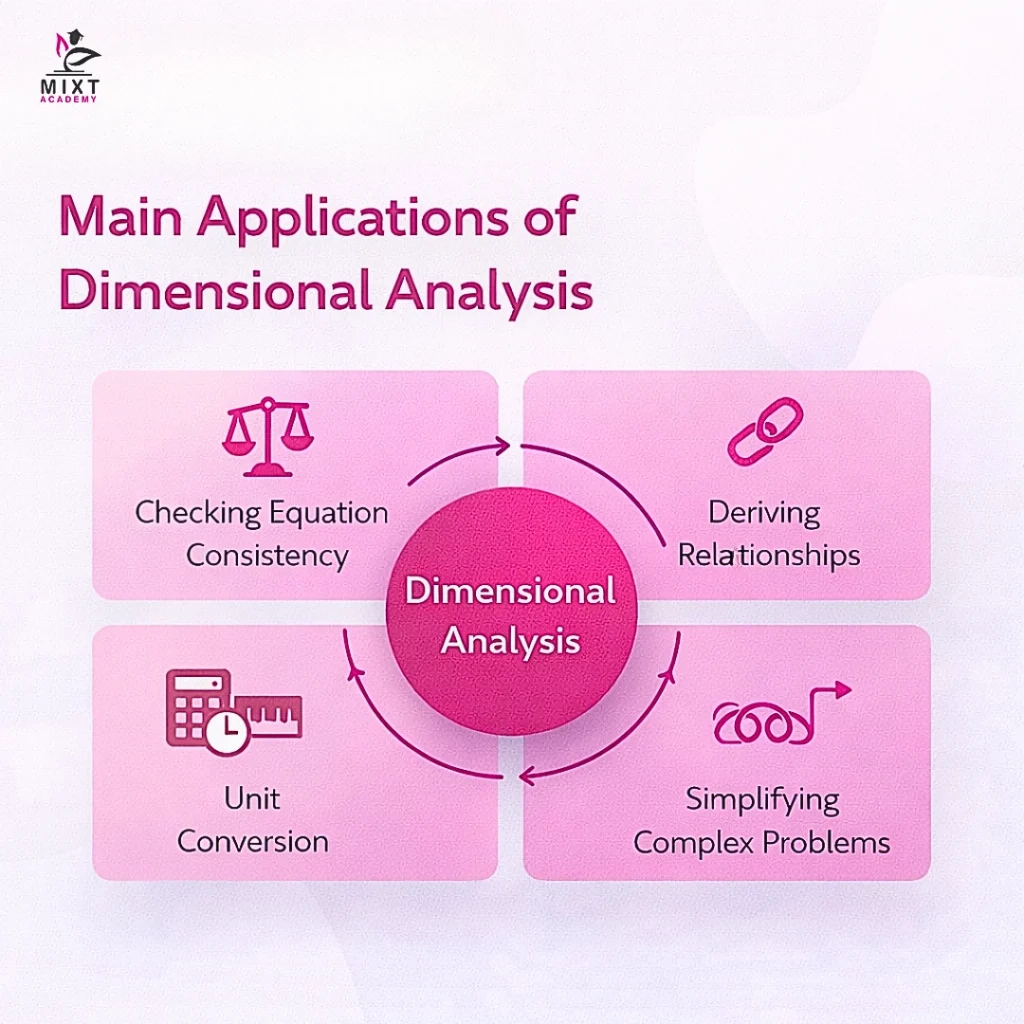
Dimensional analysis is a powerful tool in physics for understanding and working with physical quantities. It helps simplify complex problems and ensure consistency in equations. Fourier established the foundations of dimensional analysis, and it has several important applications:
- Checking Equation Consistency: Dimensional formulas can be used to verify whether a physical equation is dimensionally correct.
- Deriving Relationships: It allows us to find possible relationships between different physical quantities, even without detailed experimental data.
- Unit Conversion: Dimensional analysis provides a systematic way to convert a physical quantity from one system of units to another.
How to Check Dimensional Consistency?
- Identify all quantities in the equation and write down their dimensional formulas.
- Calculate the dimensions of each term on the left-hand side (LHS) and the right-hand side (RHS).
- Compare LHS and RHS:
- If the dimensions match, the equation is dimensionally consistent.
- If they differ, the equation is incorrect.
- Check additions and Subtractions: Only quantities with the same dimensions can be added or subtracted.
Example:
Dimensional Analysis of equation: F = m⋅a
Step 1: Left-Hand Side (LHS)
Force, F
Dimensional formula: [F] = MLT−2
Step 2: Right-Hand Side (RHS)
Mass × Acceleration: m⋅a
Mass: [m] = M
Acceleration: [a] = LT^−2
Multiply: [m][a] = M⋅LT−2 = MLT−2
Step 3: Compare
[F]_LHS = [F]_RHS = MLT^-2
Since LHS = RHS, the equation is dimensionally consistent.
Build Strong Physics Fundamentals with Units and Dimensions

Want to score better in Physics exams? Mixt Academy offers expert-led learning on Units and Dimensions, helping you master dimensional formulas, constants, and unit conversions. Our qualified tutors focus on exam-relevant concepts and problem-solving techniques that save time and reduce errors. Strengthen your Physics fundamentals with Mixt Academy. Join today and book a demo session to get started.